31. Match the compounds P-S with their carbonyl stretching frequencies (cm-1) I-VI in IR spectroscopy.
P. Acetone
I. 1870
Q. Ethyl acetate
II. 1800
R. Acetamide
III. 1740
S. Acetyl chloride
IV. 1700
V. 1660
VI. 1600
| P. Acetone | I. 1870 |
| Q. Ethyl acetate | II. 1800 |
| R. Acetamide | III. 1740 |
| S. Acetyl chloride | IV. 1700 |
| V. 1660 | |
| VI. 1600 |
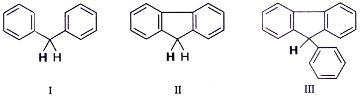
32. The decreasing order of acidity of the marked H of the following molecules is

33. On heating with dilute sulphuric acid, naphthalene-1 sulphonic acid gives predominantly
34. In the reaction, \[P{h_3}P\xrightarrow{{\,\,{\text{Mel}}\,\,}}\left[ X \right]\xrightarrow{{n - {\text{BuLi}}}}\left[ Y \right]\] the compounds X and Y, respectively, are
35. The amino acid side chain high affinity for Ca2+ and Cu2+ in metallo-proteins is
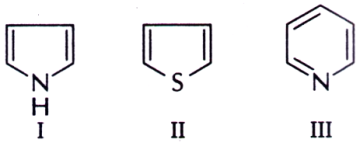
36. The decreasing order of the reactivity of the following compounds towards electrophiles is

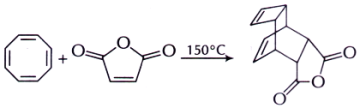
37. The two pericyclic reactions successively involved in the thermal transformation given below are

38. Bridge-head hydrogen of the conformer of cis-decalin is positioned as [a = axial; e = equatorial]
39. The compound
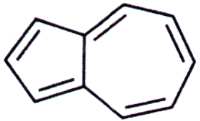
is

is
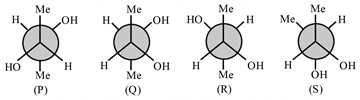
40. Among the following, the Newman projections of meso-2, 3-butanediol are

Read More Section(Basics of Organic Reaction Mechanism)
Each Section contains maximum 100 MCQs question on Basics of Organic Reaction Mechanism. To get more questions visit other sections.
