22. Which of the following statements is/are true for X?
i. X is not isoelectronic with benzene.
ii. X undergoes addition reaction with HCI.
iii. Electrophilic substitution reaction on X is much faster than that of benzene.
iv. X undergoes polymerization at 90°C
i. X is not isoelectronic with benzene.
ii. X undergoes addition reaction with HCI.
iii. Electrophilic substitution reaction on X is much faster than that of benzene.
iv. X undergoes polymerization at 90°C
23. The overall charge present on the cyclic silicate anion [Si6O18]n- is
24. Pyroxenes are class of silicate minerals, which exhibit a polymeric chain structure, as shown below
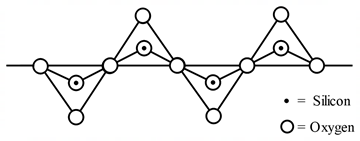
Its simplest repeating unit is

Its simplest repeating unit is
25. Using the Wade rule, the structure of B10C2H12 can be predicted. The structure and the number of isomers of B10C2H12 respectively, are
26. According to Wade's rule, the structures of B10C2H12 and [B9C2H11]2- respectively are
27. White phosphorus, P4 belongs to the
28. Match the silicate minerals (Column I) with their compositions (Column II) and order of hardness (Column III)
Column I
Column II
Column III
P. Talc
U. KAl2AlSi3O10(OH)2
X. High
Q. Muscovite
V. Mg3Si4O10(OH)2
Y. Low
R. Margarite
W. CaAl2AlSi3O10(OH)2
Z. Intermediate
| Column I | Column II | Column III |
| P. Talc | U. KAl2AlSi3O10(OH)2 | X. High |
| Q. Muscovite | V. Mg3Si4O10(OH)2 | Y. Low |
| R. Margarite | W. CaAl2AlSi3O10(OH)2 | Z. Intermediate |
