42. Which of the following is/are the examples for metal-coated articles?
43. The passive film in stainless steel forms above the
44. What is meant by vapor deposition?
45. The thickness of oxide film is 'y' at a time 't' If K1, K2 and K3 are the temperature dependent constants, the parabolic law of oxidation is given by
46. Which of the following metals is highest active in the Galvanic series?
47. A pipeline buried in soil is commonly protected from corrosion by
48. Which of the following is/are the limitations of Pourbaix diagrams?
49. Identify the false statement:
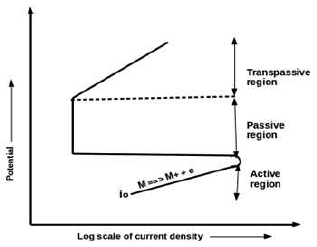
50. What is depicted in the given polarization curve?