21. The oxidation reaction lines of metals/nonmetals parallel to x- axis in Ellingham diagram is corresponds to
22. What is the temperature range for CO where it can act as reducing agent for iron oxide reduction
23. The activity of pure hydrogen gas at 1000°C and 5 atm. pressure.
24. If two system P & Q are in thermal equi librium with a third system M, then P and Q will also be in thermal equilibrium with each other. This is following . . . . . . . . law of thermodynamics.
25. As the temperature increases, the solubility of gas in molten metal . . . . . . . . at constant pressure
26. A chemical reaction whose rate does not depend on concentration of reactants is called
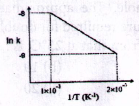
27. If k is the rate constant for a reaction and T is the absolute temperature in the given figure, the activation energy for the reaction is . . . . . . . . J/K mole.

28. The erder of a chemical reaction
29. In reaction equilibria occuring between pure condensed phases and a gas phase, the equilibrium constant, K
30. When one mole of the substance is completely burnt in oxygen. The heat evolve is known
Read More Section(Metallurgical Thermodynamics and Kinetics)
Each Section contains maximum 100 MCQs question on Metallurgical Thermodynamics and Kinetics. To get more questions visit other sections.
