A piston containing an ideal gas is originally in the state X (see figure). The gas is taken through a thermal cycle X → Y → X as shown
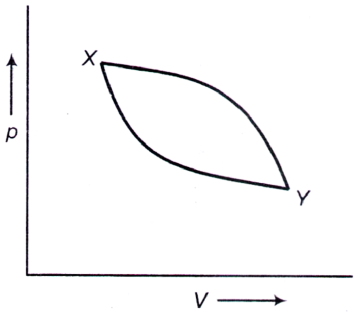
The work done by the gas is positive, if the direction of the thermal cycle is
A. clockwise
B. counter-clockwise
C. neither clockwise nor counter-clockwise
D. clockwise from X → Y and counter-clockwise from Y → X
Answer: Option A

Join The Discussion